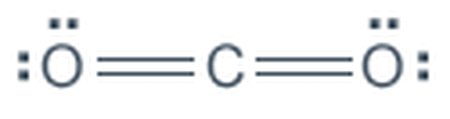Act 1 Exposition:
The molecular formula for Carbon Dioxide is CO2. The elements in carbon dioxide are two oxygen atoms covalently double bonded to one carbon atom. Carbon dioxide is made up of the elements carbon and oxygen which are both nonmetals. Carbon is found as carbon dioxide in the atmosphere and dissolved in all natural waters. There is one carbon atom and two oxygen atoms that make up carbon dioxide.
The physical properties of carbon dioxide is that it is a colorless and odorless gas at room temperature. It has a faint acid-taste and is known to also be a non-flammable gas. The solid form of carbon dioxide is usually dry ice that is white in color and can look like snowflakes or cubes in shape. Carbon dioxide can be formed during combustion, respiration, during the decomposition of organic substances, and when acids react with carbonates. The chemical properties are that it is a linear covalent molecule that can react with water to give carbonic acid or it can react with alkalis to give carbonates and bicarbonates.
Carbon dioxide is essential to life because it constantly moves from the air and is directly absorbed into water and plants through photosynthesis. It can be found in about 0.03% of air in the atmosphere. Carbon Dioxide gets released into the air again through respiration, through the decay of nature, and taking out the gases from certain water surfaces. Carbon dioxide is more dense than air and it is Earth's most significant greenhouse gas. Greenhouse gases like carbon dioxide act as a glass of a greenhouse, allowing sunlight to enter the atmosphere and warm Earth's surface without letting some of the heat from the sunlight to escape. However, carbon dioxide is leading to global warming because of the increase in Earth's average temperature. Some examples of how carbon dioxide can be used are as dry ice and carbonated drinks. It can also be used to preserve rocks for geological means by injecting some carbon dioxide into the atmosphere as well as to produce biomass energy and combust fossil fuels. Some scientists think that fossils fuels are the reason that carbon dioxide's atmospheric concentration is rising. It can be found in solution such as in water lakes, streams, ponds, and oceans.
Some of the different names of CO2 are carbon dioxide, fixed air, carbonic acid gas, and carbonic anhydride. Carbonic acid gas can be classified as inorganic because carbon is not covalently bonded to hydrogen or other elements; it only consists of carbon hence it is not organic.
The formula weight of carbon dioxide (CO2) is 44.0095g/mol.
Even though there are two oxygens for every one carbon in carbon dioxide, most of the mass of carbon dioxide is due to the oxygen. This is found by calculating the mass % of each element.
% oxygen = 36.4%
I found this by dividing the mass of oxygen (16.00g/mol) by the molar mass of carbon dioxide (44.0095g/mol) and multiplying it by 100%.
% carbon = 27.3%
I found this by dividing the mass of carbon (12.01g/mol) by the molar mass of carbon dioxide (44.0095g/mol) and multiplying it by 100%.
The molecular formula for Carbon Dioxide is CO2. The elements in carbon dioxide are two oxygen atoms covalently double bonded to one carbon atom. Carbon dioxide is made up of the elements carbon and oxygen which are both nonmetals. Carbon is found as carbon dioxide in the atmosphere and dissolved in all natural waters. There is one carbon atom and two oxygen atoms that make up carbon dioxide.
The physical properties of carbon dioxide is that it is a colorless and odorless gas at room temperature. It has a faint acid-taste and is known to also be a non-flammable gas. The solid form of carbon dioxide is usually dry ice that is white in color and can look like snowflakes or cubes in shape. Carbon dioxide can be formed during combustion, respiration, during the decomposition of organic substances, and when acids react with carbonates. The chemical properties are that it is a linear covalent molecule that can react with water to give carbonic acid or it can react with alkalis to give carbonates and bicarbonates.
Carbon dioxide is essential to life because it constantly moves from the air and is directly absorbed into water and plants through photosynthesis. It can be found in about 0.03% of air in the atmosphere. Carbon Dioxide gets released into the air again through respiration, through the decay of nature, and taking out the gases from certain water surfaces. Carbon dioxide is more dense than air and it is Earth's most significant greenhouse gas. Greenhouse gases like carbon dioxide act as a glass of a greenhouse, allowing sunlight to enter the atmosphere and warm Earth's surface without letting some of the heat from the sunlight to escape. However, carbon dioxide is leading to global warming because of the increase in Earth's average temperature. Some examples of how carbon dioxide can be used are as dry ice and carbonated drinks. It can also be used to preserve rocks for geological means by injecting some carbon dioxide into the atmosphere as well as to produce biomass energy and combust fossil fuels. Some scientists think that fossils fuels are the reason that carbon dioxide's atmospheric concentration is rising. It can be found in solution such as in water lakes, streams, ponds, and oceans.
Some of the different names of CO2 are carbon dioxide, fixed air, carbonic acid gas, and carbonic anhydride. Carbonic acid gas can be classified as inorganic because carbon is not covalently bonded to hydrogen or other elements; it only consists of carbon hence it is not organic.
The formula weight of carbon dioxide (CO2) is 44.0095g/mol.
Even though there are two oxygens for every one carbon in carbon dioxide, most of the mass of carbon dioxide is due to the oxygen. This is found by calculating the mass % of each element.
% oxygen = 36.4%
I found this by dividing the mass of oxygen (16.00g/mol) by the molar mass of carbon dioxide (44.0095g/mol) and multiplying it by 100%.
% carbon = 27.3%
I found this by dividing the mass of carbon (12.01g/mol) by the molar mass of carbon dioxide (44.0095g/mol) and multiplying it by 100%.
the types of bonds found in water are polar covalent.
Atoms Difference in Electronegativity Type of Bond
O-C 3.5-2.5=1.0 Polar Covalent
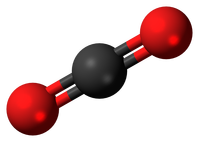
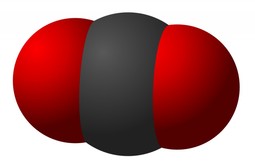
models of carbon dioxide
Space-filling
References:
CRC Handbook:
http://www.fptl.ru/biblioteka/spravo4niki/handbook-of-Chemistry-and-Physics.pdf
Chemistry Textbook: A Molecular Approach by Nivaldo J. Tro
Chemical Propeties of Carbon Dioxide:
http://www.ucc.ie/academic/chem/dolchem/html/comp/co2.html
Where is Carbon Dioxide Found:
http://water.me.vccs.edu/exam_prep/carbondioxide.html
"Carbon Dioxide, Which Has the Molecular Formula CO2." Government of Canada, Environment Canada. <https://www.ec.gc.ca/toxiques-toxics/Default.asp?lang=En&n=98E80CC6-1&xml=DF76322C-49E3-4335-811F-D0E1DA54938A>.
Carbon Dioxide. http://water.me.vccs.edu/exam_prep/carbondioxide.html
Wisconsin Department of Health Services. Carbon Dioxide. https://www.dhs.wisconsin.gov/chemical/carbondioxide.htm
PubChem Open Chemistry Database. http://pubchem.ncbi.nlm.nih.gov/compound/carbon_dioxide#section=Wikipedia
Carbon Dioxide Electron/Lewis Dot Structure Image. http://www.wiredchemist.com/chemistry/instructional/an-introduction-to-chemistry/structure/the-valence-shell-electron-pair-repulsion-model
Carbon Dioxide Ball and Stick Image. http://upload.wikimedia.org/wikipedia/commons/a/a0/Carbon_dioxide_3D_ball.png
Carbon Dioxide Space-Filling Image. http://doc-snow.hubpages.com/hub/Water-Is-A-Dancer#slide4266684
Covalent Bonds and Other Bonds. https://www.boundless.com/biology/textbooks/boundless-biology-textbook/the-chemical-foundation-of-life-2/atoms-isotopes-ions-and-molecules-the-building-blocks-50/covalent-bonds-and-other-bonds-and-interactions-280-11413/images/fig-ch02_01_11/
http://www.fptl.ru/biblioteka/spravo4niki/handbook-of-Chemistry-and-Physics.pdf
Chemistry Textbook: A Molecular Approach by Nivaldo J. Tro
Chemical Propeties of Carbon Dioxide:
http://www.ucc.ie/academic/chem/dolchem/html/comp/co2.html
Where is Carbon Dioxide Found:
http://water.me.vccs.edu/exam_prep/carbondioxide.html
"Carbon Dioxide, Which Has the Molecular Formula CO2." Government of Canada, Environment Canada. <https://www.ec.gc.ca/toxiques-toxics/Default.asp?lang=En&n=98E80CC6-1&xml=DF76322C-49E3-4335-811F-D0E1DA54938A>.
Carbon Dioxide. http://water.me.vccs.edu/exam_prep/carbondioxide.html
Wisconsin Department of Health Services. Carbon Dioxide. https://www.dhs.wisconsin.gov/chemical/carbondioxide.htm
PubChem Open Chemistry Database. http://pubchem.ncbi.nlm.nih.gov/compound/carbon_dioxide#section=Wikipedia
Carbon Dioxide Electron/Lewis Dot Structure Image. http://www.wiredchemist.com/chemistry/instructional/an-introduction-to-chemistry/structure/the-valence-shell-electron-pair-repulsion-model
Carbon Dioxide Ball and Stick Image. http://upload.wikimedia.org/wikipedia/commons/a/a0/Carbon_dioxide_3D_ball.png
Carbon Dioxide Space-Filling Image. http://doc-snow.hubpages.com/hub/Water-Is-A-Dancer#slide4266684
Covalent Bonds and Other Bonds. https://www.boundless.com/biology/textbooks/boundless-biology-textbook/the-chemical-foundation-of-life-2/atoms-isotopes-ions-and-molecules-the-building-blocks-50/covalent-bonds-and-other-bonds-and-interactions-280-11413/images/fig-ch02_01_11/