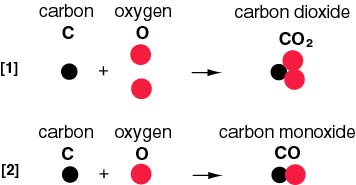
Carbon dioxide has similar properties to carbon monoxide. The main difference is that carbon dioxide contains another oxygen atom so if there is insufficient oxygen, carbon monoxide is formed instead (as shown in the picture to the left). Carbon dioxide is the gas we exhale while carbon monoxide is a highly poisonous gas. Carbon monoxide is a neutral oxide that can burn in the air to give carbon dioxide. They have similar properties because they are almost the same molecule except one has another oxygen while the other does not. According to our Chemistry textbook, the presence of the second oxygen in carbon dioxide prevents it from binding to the oxygen-carrying site in hemoglobin, making it far less toxic than carbon monoxide. Although high levels of carbon dioxide can be hazardous for other reasons, lower levels can enter the bloodstream with no adverse effects.
Carbon plays the role of an atom that has an incomplete outermost electron shell. The atomic number of carbon is six which means it has six protons and electrons, the first two fill the inner shell, leaving four in the second shell. Two electrons fill the inner shell and leaves four in the second shell. Carbon atoms can form four covalent bonds with other atoms to satisfy the octet rule. An example would be methane since its chemical formula is CH4.
Oxygen makes sure that it helps the process of creating energy in living cells. Plants, animals, and living things need oxygen to survive. Oxygen is cycled through the environment through carbon dioxide so that plants are able to use the energy. Plants thus convert sunlight carbon dioxide and water into carbohydrates and oxygen which results in photosynthesis. This means that carbon dioxide breathes in carbon dioxide and breathes out oxygen. Humans and animals also breathe in oxygen to break down carbohydrates into energy which results in respiration.
An isomer of carbon dioxide is carbon trioxide. It can be formed in low temperature carbon dioxide matrix. It is in carbon-dioxide-rich extraterrestrial ices.
A new two-dimensional polymer, PG-ES1 allows for highly efficient carbon dioxide separation. It has been created by Department of Energy’s National Energy Research Scientific Computing Center (NERSC). PG-ES1 is more permeable to carbon dioxide than other existing materials. It maintains the rejection of nitrogen and methane gases. It acts as a molecular filter that allows carbon dioxide to pass easily while not letting other gases escape.
Carbon dioxide does not contain organic end groups because it is inorganic.
Carbon plays the role of an atom that has an incomplete outermost electron shell. The atomic number of carbon is six which means it has six protons and electrons, the first two fill the inner shell, leaving four in the second shell. Two electrons fill the inner shell and leaves four in the second shell. Carbon atoms can form four covalent bonds with other atoms to satisfy the octet rule. An example would be methane since its chemical formula is CH4.
Oxygen makes sure that it helps the process of creating energy in living cells. Plants, animals, and living things need oxygen to survive. Oxygen is cycled through the environment through carbon dioxide so that plants are able to use the energy. Plants thus convert sunlight carbon dioxide and water into carbohydrates and oxygen which results in photosynthesis. This means that carbon dioxide breathes in carbon dioxide and breathes out oxygen. Humans and animals also breathe in oxygen to break down carbohydrates into energy which results in respiration.
An isomer of carbon dioxide is carbon trioxide. It can be formed in low temperature carbon dioxide matrix. It is in carbon-dioxide-rich extraterrestrial ices.
A new two-dimensional polymer, PG-ES1 allows for highly efficient carbon dioxide separation. It has been created by Department of Energy’s National Energy Research Scientific Computing Center (NERSC). PG-ES1 is more permeable to carbon dioxide than other existing materials. It maintains the rejection of nitrogen and methane gases. It acts as a molecular filter that allows carbon dioxide to pass easily while not letting other gases escape.
Carbon dioxide does not contain organic end groups because it is inorganic.
References:
Chemistry Textbook: A Molecular Approach by Nivaldo J. Tro
Carbon Monoxide: http://www.ucc.ie/academic/chem/dolchem/html/comp/co.html
The importance of Carbon: https://www.boundless.com/biology/textbooks/boundless-biology-textbook/the-chemical-foundation-of-life-2/carbon-52/the-importance-of-carbon-288-11421/
The Oxygen Cycle: http://water.me.vccs.edu/concepts/oxycycle.html
Oxygen and Carbon Dioxide: http://www.miseagrant.umich.edu/lessons/lessons/by-broad-concept/earth-science/water-quality/oxygen-and-carbon-dioxide/
Cyclic Carbon Trioxide Isomer: http://pubs.rsc.org/en/Content/ArticleLanding/2004/CP/b315626p#!divAbstract
Researchers create carbon-dioxide separating polymer: http://phys.org/news/2012-08-carbon-dioxide-separating-polymer.html
http://www.chemistryexplained.com/Ny-Pi/Organic-Chemistry.html
Chemistry Textbook: A Molecular Approach by Nivaldo J. Tro
Carbon Monoxide: http://www.ucc.ie/academic/chem/dolchem/html/comp/co.html
The importance of Carbon: https://www.boundless.com/biology/textbooks/boundless-biology-textbook/the-chemical-foundation-of-life-2/carbon-52/the-importance-of-carbon-288-11421/
The Oxygen Cycle: http://water.me.vccs.edu/concepts/oxycycle.html
Oxygen and Carbon Dioxide: http://www.miseagrant.umich.edu/lessons/lessons/by-broad-concept/earth-science/water-quality/oxygen-and-carbon-dioxide/
Cyclic Carbon Trioxide Isomer: http://pubs.rsc.org/en/Content/ArticleLanding/2004/CP/b315626p#!divAbstract
Researchers create carbon-dioxide separating polymer: http://phys.org/news/2012-08-carbon-dioxide-separating-polymer.html
http://www.chemistryexplained.com/Ny-Pi/Organic-Chemistry.html
