|
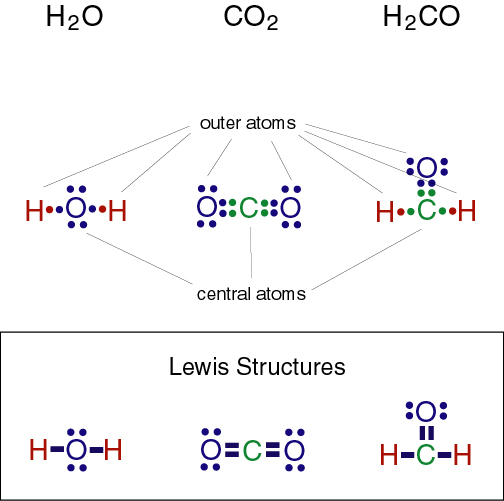
The Lewis Dot Structure of carbon dioxide.
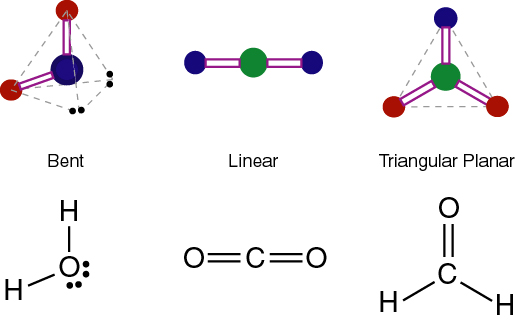
The linear geometry of carbon dioxide.
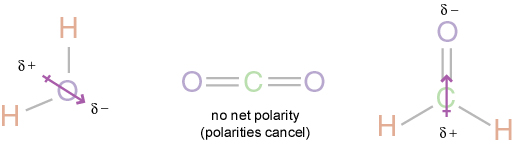
This image shows how the net polarities cancel out due to similar electronegativities of 0.89 from thee oxygen atoms.
|
Carbon Dioxide is nonpolar. To determine the polarity of Carbon Dioxide, one must look at the Lewis Dot structure first. The Lewis Dot structure helps look at whether the molecule has 2 or more atoms and whether it has central or outer atoms. Determining the shape helps to look at molecules with three or more atoms. Using the shape of molecule and bond polarities, one can determine the polarity of the molecule. The two oxygens in Carbon Dioxide each have the electronegativity of 3.44 while Carbon is 2.55. The electronegativity tells that the Oxygens pull more strongly on the electrons in Carbon Dioxide. The difference in electronegativity is 0.89 so the electrons are equally balanced between the two Oxygen atoms. The polarities cancel out, making Carbon Dioxide nonpolar.
Carbon Dioxide has polar bonds because of the individual electronegativities of carbon and oxygen. The geometry of carbon dioxide is linear. Carbon dioxide has linear geometry because it has two electron groups and no lone pairs to affect the orientation of the molecules.The linear orientation reduces the repulsion forces. The polar bonds cancel which makes the molecule nonpolar. Since it is nonpolar, it does not have dipole-dipole forces. Carbon dioxide does not form a hydrogen bond because it does not have any hydrogen atoms. Carbon dioxide forms the intermolecular bond of Van der Waals. It is caused by dipoles from movements of electrons. The movements attract other molecules by causing similar dipoles in them. Carbon Dioxide is very stable which means it causes it to make more effort for the molecules to activate and react. A chemical reaction of carbon dioxide is when it reacts with lithium nitride. The reaction forms carbon nitride and lithium cyanide, making carbon dioxide into a solid. Carbon dioxide can also react with water to form a weak acid. The reaction between carbon dioxide and water can be observed by using and acid-base indicator to see whether it will change color. Carbon dioxide is an acidic oxide that when it reacts with water, it gives carbonic acid. When carbon dioxides react with alkalis, it gives carbonates and bicarbonates. |
Carbon Dioxide reacts with Lithium Nitride to form Carbon Nitride and Lithium Cyanide:
CO2 + Li3N ----> C3N4 + Li2CN2
Carbon dioxide reacts with water to give carbonic acid:
CO2 + H2O ----> H2CO3
Carbon dioxide reacts with alkalis to give carbonates or bicarbonates:
CO2 + NaOH ----> NaHCO3 Sodium Bicarbonate
NaHCO3 + NaOH ----> Na2CO3 (Sodium Carbonate) + H2O
Carbon Dioxide as a Limiting Reactant
Carbon dioxide reacts with water to give carbonic acid:
CO2 + H2O ----> H2CO3
100gCO2 * 1 mol CO2 * 1 molH2CO3 = 2.27 mol H2CO3
44gCO2 1molCO2
100gH2O * 1mol H2O * 1molH2CO3 = 5.56 mol H2CO3
18gH2O 1mol H2O
Carbon dioxide is the limiting reactant since it produced the least product which is carbonic acid.
CO2 + Li3N ----> C3N4 + Li2CN2
Carbon dioxide reacts with water to give carbonic acid:
CO2 + H2O ----> H2CO3
Carbon dioxide reacts with alkalis to give carbonates or bicarbonates:
CO2 + NaOH ----> NaHCO3 Sodium Bicarbonate
NaHCO3 + NaOH ----> Na2CO3 (Sodium Carbonate) + H2O
Carbon Dioxide as a Limiting Reactant
Carbon dioxide reacts with water to give carbonic acid:
CO2 + H2O ----> H2CO3
100gCO2 * 1 mol CO2 * 1 molH2CO3 = 2.27 mol H2CO3
44gCO2 1molCO2
100gH2O * 1mol H2O * 1molH2CO3 = 5.56 mol H2CO3
18gH2O 1mol H2O
Carbon dioxide is the limiting reactant since it produced the least product which is carbonic acid.
References:
Determining Polarity of Molecules: http://www.marin.edu/homepages/ErikDunmire/CHEM105/Concept_Review/Polarity/Polarity.html
UC Davis Chemwiki: http://chemwiki.ucdavis.edu/Theoretical_Chemistry/Chemical_Bonding/Lewis_Theory_of_Bonding/Geometry_of_Molecules
Intermolecular bonds of CO2 : http://socratic.org/questions/what-type-of-intermolecular-force-of-attraction-is-found-in-co2-2
Chemical & Engineering News: http://cen.acs.org/articles/85/i18/Carbon-Dioxide.html
Chemical Reaction: http://www.gizmag.com/co2-li3n-reaction/22620/
Carbon Dioxide: http://www.ucc.ie/academic/chem/dolchem/html/comp/co2.html
Determining Polarity of Molecules: http://www.marin.edu/homepages/ErikDunmire/CHEM105/Concept_Review/Polarity/Polarity.html
UC Davis Chemwiki: http://chemwiki.ucdavis.edu/Theoretical_Chemistry/Chemical_Bonding/Lewis_Theory_of_Bonding/Geometry_of_Molecules
Intermolecular bonds of CO2 : http://socratic.org/questions/what-type-of-intermolecular-force-of-attraction-is-found-in-co2-2
Chemical & Engineering News: http://cen.acs.org/articles/85/i18/Carbon-Dioxide.html
Chemical Reaction: http://www.gizmag.com/co2-li3n-reaction/22620/
Carbon Dioxide: http://www.ucc.ie/academic/chem/dolchem/html/comp/co2.html