Carbon dioxide is used:
Industry
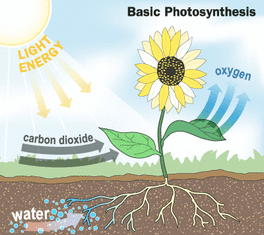
Carbon Dioxide is found in the air and in water as a part of the carbon cycle, which means that it can be used/produced both in the factory and in nature. It is on the most abundant gases in the atmosphere and is vital in the plant and animal process such as photosynthesis and respiration. Photosynthesis and respiration play an important role in the carbon cycle and are at equilibrium with one another. Photosynthesis dominates during the warm part of the year, while respiration in the colder part.
According to the Merck Manual, carbon dioxide is maintained between the pressures of 35 and 45 mmHg. Abnormal PCO2 is linked to disorders of ventilation and is associated with acid-base changes. Carbon Dioxide can cause Hypercapnia, Hypocapnia, Carboxyhemoglobinemia, and Methemoglobinemia.
Carbon Dioxide is significant because it can be used in the industry, environment, and to improve health. In history, it played as a way of photosynthesis, respiration, and the greenhouse effect just as it does today. Scottish chemist Joseph Black discovered Carbon Dioxide on June 11th, 1754. Black noticed that when heating, calcium carbonate (CaCO3) produced a gas that is denser than air and that it could not sustain fire or animal life. He called it “fixed air” which is known as Carbon Dioxide today.
Plants can take in carbon dioxide and use it to make sugars, other than that, carbon dioxide is not used to make other molecules.
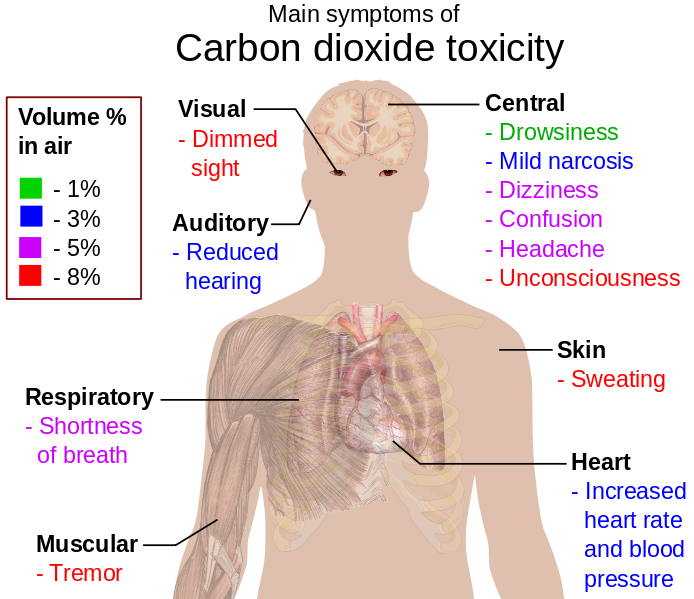
Carbon dioxide helps humans breathe as well as in photosynthesis and respiration. Normal levels of carbon dioxide are harmless, but under the right conditions, it can still cause adverse effects. High concentrations in confined areas can be dangerous and act as an oxygen displacer. It can cause reactions such as dizziness, disorientation, suffocation, and even death. Death occurs when there is a depression of the central nervous system with high exposure to carbon dioxide. Some other symptoms from low to high concentrations depend on the percent of carbon dioxide they are exposed to. About 2 to 3% can cause shortness of breath and deep breathing. About 7.5% causes headaches, dizziness, restlessness, breathlessness, increased heart rate, and visual distortion. At 30% it can cause coma, convulsions, and death. Such exposure can also occur in occupations such as mining, brewing, and submarine workers.
Carbon dioxide warms the Earth because it is a greenhouse gas in the atmosphere. It also causes plants to provide less evaporative cooling. Trees and other plants keep the planet cool, but rising levels of carbon dioxide are turning down this global air conditioner.Trees which sequester carbon, die off, and release their stores CO2. However, they are not a permanent solution to global warming. Plants absorb carbon dioxide for photosynthesis in pores called stomata. When the carbon dioxide levels are high, the leaf pores shrink, causing less water to be released and diminishing the tree’s coolin power. Scientists are concerned that carbon dioxide is decreasing the evaporative cooling by plants and adds to global warming. Scientists say that how plants respond to carbon dioxide is very important for making good climate predictions.
Industry
- by food industries to produce carbonated beverages such as mineral water and sodas (picture shown to the left).
- as a foam creator in beer or as inerting atmosphere.
- as an inerting atmosphere such as for wielding or handling flammable materials.
- It can create a protective atmosphere that allows to control micro-organisms such as larvae, bacteria, and fungi.
- in fire extinguishers. It is packaged in a gaseous state which turns into dry ice snow when released. It can cool flames and deprives fire of the oxygen it burns by displacing air.
- At 78 degrees Celsius, in a solid state, Carbon dioxide can form dry ice. It is used to transport food that the cold chain is not broken. Meals are kept safe and fresh for consumption.
- the PH level can be controlled by injecting Carbon Dioxide, such as in the treatment of waste water. This method is another alternative for sulfuric acid.
- More than 90% of dry matter (biomass) produced by plants come from assimilated CO (Book reference).
- Dry ice is used to transport and preserve tissues at low temperatures, which is necessary for transplants.
- In a gaseous state, it helps create an atmosphere that is close to physiological conditions.
- Develops modern mini-invasive surgical procedures.
Carbon Dioxide is found in the air and in water as a part of the carbon cycle, which means that it can be used/produced both in the factory and in nature. It is on the most abundant gases in the atmosphere and is vital in the plant and animal process such as photosynthesis and respiration. Photosynthesis and respiration play an important role in the carbon cycle and are at equilibrium with one another. Photosynthesis dominates during the warm part of the year, while respiration in the colder part.
According to the Merck Manual, carbon dioxide is maintained between the pressures of 35 and 45 mmHg. Abnormal PCO2 is linked to disorders of ventilation and is associated with acid-base changes. Carbon Dioxide can cause Hypercapnia, Hypocapnia, Carboxyhemoglobinemia, and Methemoglobinemia.
Carbon Dioxide is significant because it can be used in the industry, environment, and to improve health. In history, it played as a way of photosynthesis, respiration, and the greenhouse effect just as it does today. Scottish chemist Joseph Black discovered Carbon Dioxide on June 11th, 1754. Black noticed that when heating, calcium carbonate (CaCO3) produced a gas that is denser than air and that it could not sustain fire or animal life. He called it “fixed air” which is known as Carbon Dioxide today.
Plants can take in carbon dioxide and use it to make sugars, other than that, carbon dioxide is not used to make other molecules.
Carbon dioxide helps humans breathe as well as in photosynthesis and respiration. Normal levels of carbon dioxide are harmless, but under the right conditions, it can still cause adverse effects. High concentrations in confined areas can be dangerous and act as an oxygen displacer. It can cause reactions such as dizziness, disorientation, suffocation, and even death. Death occurs when there is a depression of the central nervous system with high exposure to carbon dioxide. Some other symptoms from low to high concentrations depend on the percent of carbon dioxide they are exposed to. About 2 to 3% can cause shortness of breath and deep breathing. About 7.5% causes headaches, dizziness, restlessness, breathlessness, increased heart rate, and visual distortion. At 30% it can cause coma, convulsions, and death. Such exposure can also occur in occupations such as mining, brewing, and submarine workers.
Carbon dioxide warms the Earth because it is a greenhouse gas in the atmosphere. It also causes plants to provide less evaporative cooling. Trees and other plants keep the planet cool, but rising levels of carbon dioxide are turning down this global air conditioner.Trees which sequester carbon, die off, and release their stores CO2. However, they are not a permanent solution to global warming. Plants absorb carbon dioxide for photosynthesis in pores called stomata. When the carbon dioxide levels are high, the leaf pores shrink, causing less water to be released and diminishing the tree’s coolin power. Scientists are concerned that carbon dioxide is decreasing the evaporative cooling by plants and adds to global warming. Scientists say that how plants respond to carbon dioxide is very important for making good climate predictions.
References:
Merck Manual:
http://dams.insomnia247.nl/Public/Medical/Medicine/The%20Merck%20Manual%20of%20Diagnosis%20and%20Therapy%2019th%20Edition.pdf
Hope for A Heated Planet (Book Reference):
http://search.ebscohost.com/login.aspx?direct=true&db=nlebk&AN=263603&site=ehost-live&ebv=EB&ppid=pp_188
Carbon Dioxide and Environmental Stress
http://search.ebscohost.com/login.aspx?direct=true&db=nlebk&AN=203241&site=ehost-live&ebv=EB&ppid=pp_4
Carbon Dioxide Applications:
http://www.airliquide.com/en/company/our-businesses-our-products/other-gases/carbon-dioxide-10/carbon-dioxide-applications.html
Carbon Dioxide:
http://www.lenntech.com/carbon-dioxide.htm
Learn Chemistry:
http://www.rsc.org/learn-chemistry/resource/rdc00000611/on-this-day-jun-11-carbon-dioxide-discovered?cmpid=CDC00000611
Health Risk Evaluation for Carbon Dioxide:
http://www.blm.gov/style/medialib/blm/wy/information/NEPA/cfodocs/howell.Par.2800.File.dat/25apxC.pdf
Carnegie Institution. "Carbon dioxide's effects on plants increase global warming, study finds." ScienceDaily. ScienceDaily, 4 May 2010. <www.sciencedaily.com/releases/2010/05/100503161435.htm>.
Image from:
https://www.google.com/search?q=carbon+dioxide+soluble+in+water&espv=2&biw=1366&bih=667&source=lnms&tbm=isch&sa=X&ei=DupjVfjpBIOeNurCgEA&sqi=2&ved=0CAgQ_AUoAw#tbm=isch&q=carbon+dioxide+in+sodas&imgrc=sDoyP-gz1Kq_nM%253A%3BjZrnIWXaXIhS6M%3Bhttp%253A%252F%252Fwww.thenakedscientists.com%252FHTML%252Fuploads%252FRTEmagicC_ExplodingCoke-opened_02.gif.gif%3Bhttp%253A%252F%252Fwww.thenakedscientists.com%252FHTML%252Fexperiments%252Fexp%252Fexploding-coke%252F%3B150%3B337
Merck Manual:
http://dams.insomnia247.nl/Public/Medical/Medicine/The%20Merck%20Manual%20of%20Diagnosis%20and%20Therapy%2019th%20Edition.pdf
Hope for A Heated Planet (Book Reference):
http://search.ebscohost.com/login.aspx?direct=true&db=nlebk&AN=263603&site=ehost-live&ebv=EB&ppid=pp_188
Carbon Dioxide and Environmental Stress
http://search.ebscohost.com/login.aspx?direct=true&db=nlebk&AN=203241&site=ehost-live&ebv=EB&ppid=pp_4
Carbon Dioxide Applications:
http://www.airliquide.com/en/company/our-businesses-our-products/other-gases/carbon-dioxide-10/carbon-dioxide-applications.html
Carbon Dioxide:
http://www.lenntech.com/carbon-dioxide.htm
Learn Chemistry:
http://www.rsc.org/learn-chemistry/resource/rdc00000611/on-this-day-jun-11-carbon-dioxide-discovered?cmpid=CDC00000611
Health Risk Evaluation for Carbon Dioxide:
http://www.blm.gov/style/medialib/blm/wy/information/NEPA/cfodocs/howell.Par.2800.File.dat/25apxC.pdf
Carnegie Institution. "Carbon dioxide's effects on plants increase global warming, study finds." ScienceDaily. ScienceDaily, 4 May 2010. <www.sciencedaily.com/releases/2010/05/100503161435.htm>.
Image from:
https://www.google.com/search?q=carbon+dioxide+soluble+in+water&espv=2&biw=1366&bih=667&source=lnms&tbm=isch&sa=X&ei=DupjVfjpBIOeNurCgEA&sqi=2&ved=0CAgQ_AUoAw#tbm=isch&q=carbon+dioxide+in+sodas&imgrc=sDoyP-gz1Kq_nM%253A%3BjZrnIWXaXIhS6M%3Bhttp%253A%252F%252Fwww.thenakedscientists.com%252FHTML%252Fuploads%252FRTEmagicC_ExplodingCoke-opened_02.gif.gif%3Bhttp%253A%252F%252Fwww.thenakedscientists.com%252FHTML%252Fexperiments%252Fexp%252Fexploding-coke%252F%3B150%3B337